 |
Cleveland Clinic announces Top Ten medical innovations for 2015, mobile stroke unit and Dengue vaccine top list (video) |
CLEVELAND, Ohio -- A high-tech $1 million ambulance-like mobile unit that can diagnose and treat stroke in the field is the top medical innovation for 2015 atCleveland Clinic's annual Medical Innovation Summit.
A panel of Clinic physicians unveiled the highly anticipated top 10 list today to the roughly 1,500 doctors, entrepreneurs and industry leaders attending the 12th annual summit in downtown Cleveland at the Convention Center. The top medical innovations are expected to have the biggest impacts on medicine within the coming year.
Among the top ten innovations are a first-ever vaccine for Dengue fever, a fast and painless new way to draw blood, and a tiny wireless pacemaker no bigger than a AAA battery.
More than 150 Clinic staff members each made several nominations for the list in the spring, and expert committees within the health system pared down the selections.
Dr. Michael Roizen, chief medical officer of the Clinic's Wellness Institute and a practicing anesthesiologist and internist, said the Clinic's Top Ten list has a pretty good track record at predicting big breakthroughs that will have a direct impact on patients.
"If you look two years back, almost all of them have made it to clinical practice and made a significant difference," Roizen told The Plain Dealer. The selections are a gamble, he said, because of guesses in terms of the regulatory process and the outcome of ongoing studies.
"We're often 6 months to 18 months early, but the predictions are often borne out," he said.
The mobile stroke unit, which the Clinic launched in late May, is the only innovation on the list for which the Clinic was the primary researcher or developer. It earned the top spot due to how much faster patients using the unit are receiving treatment, which became obvious very quickly, Roizen said.
"What turned the tide on that one was the data, without a doubt," he said.
This year's Top Ten Innovations are:
1. Mobile stroke ambulance
In May, the Cleveland Cliniclaunched its $1 million mobile stroke unit, an ambulance-like vehicle that is equipped with a CT scan, a broadband telemedicine link to Clinic neurologists who can provide a rapid stroke diagnosis, and life-saving drugs that can open up blood vessels in the brain and prevent damage.
The unit, which is based at the Clinic and has treated between 80 and 90 patients so far, launches in tandem with Cleveland EMS after 911 calls for suspected stroke in the city of Cleveland. So far, data show that patients are getting life-saving therapy twice as often as they would using traditional methods, and four times as fast, Dr. Peter Rasmussen, director of the Clinic's Cerebrovascular Center.
"In my opinion there is no better place to be managed [for stroke] in this world than on the back of that ambulance or treatment unit," he said.
Stroke, which happens when blood flow to part of the brain is blocked (called ischemic stroke) or when sudden bleeding damages brain cells (called hemorrhagic stroke), is the fourth-leading cause of death and a leading cause of serious, long-term disability in the United States.
The only FDA-approved treatment for ischemic strokes, which account for about 87 percent of all strokes, is a clot-busting therapy called tPA, or tissue plasminogen activator. The drug, administered by IV, has to be delivered quickly — within three hours ideally. Patients with ischemic stroke who receive tPA within an hour are twice as likely to recover with good function as those who receive the drug within a six-hour window.
Since the launch of the mobile unit, patients are receiving tPA on average within 19 minutes of arrival of the team, Rasmussen said. That means the patient has had a CT scan and blood tests on board, the scan has been reviewed remotely by a Clinic doctor, and tPA has been started in the vehicle, all before arrival in the hospital. One patient received the drug within 11 minutes.
"Even Cleveland Clinic's main campus can't even begin to compete with those numbers," Rasmussen said. The average tPA delivery time on the main campus is about 60 minutes.
The Clinic's unit is based on a similar model at Charité-Universitätsmedizin Berlin, that city's teaching hospital, where a mobile stroke unit began treating patients within a clinical trial setting in 2011. The Clinic's unit is currently only active 12 hours a day, from 8 a.m. to 8 p.m., but Rasmussen said the team is looking into extending hours. They are called on an average of four or five possible strokes a day.
2. Dengue vaccine
The first vaccine against Dengue fever, a painful mosquito-borne virus common in Africa and Asia, is now very close to commercial availability after completing large-scale clinical trials this fall.
The vaccine, which is being developed by French drug company Sanofi, has shown great promise in protecting children from the disease: the vaccine cut the risk of getting the disease by 60.8 percent the most recent clinical trial, which involved more than 20,000 children ages 9 to 16 from several countries in Latin America and the Caribbean. Kids who received the vaccine also had an 80.3 percent lower risk of being hospitalized for dengue compared with those who received a placebo.
Dengue fever infects as many as 400 million people yearly, according to the Centers for Disease Control and Prevention (CDC). Although the disease rarely occurs in the U.S., it has recently been spreading to previously unaffected areas of Japan, India and the Pacific Islands, and is endemic in Puerto Rico. It has also been reported in small outbreaks in Florida and Texas.
Sanofi officials have said they could apply for approval of the vaccine in the first quarter of 2015.
3. New art of blood collection/diagnosis
A Palo Alto-based company's big idea for blood testing is very small: Their labs can run a bevvy of diagnostic tests on a single drop of blood.
Theranos' new method for blood collection, which involves no needles, takes a micro sample of blood from capillaries in the finger and stores it in a small container for analysis at one of the company's two labs. The company does not reveal its proprietary techniques.
The goal of the technique is to reduce pain, time, cost, and errors of conventional blood testing. The company can perform up to 30 tests on a single sample, and provides a complete list of the tests available, along with their prices.
A standard lipid panel, for example, will cost $2.99, and can cost anywhere from $60 to hundreds of dollars, depending on where and when it's performed.
4. PCSK9-inhibitors for cholesterol reduction
A new class of injectable cholesterol-lowering medications which arose from research into a rare genetic disorder, the PCSK9-inhibitors are being hailed as the biggest breakthrough for the treatment of heart disease since the discovery of statins.
Doctors use statins, which include familiar brand names such as Lipitor, Crestor, Vytorin and Zocor, to lower LDL or "bad" cholesterol in about 25 million patients in the U.S. every year. They are very effective both at reducing symptoms and cutting down the risk of death, but not for everyone.
Up to 20 percent of people who take the drugs deemed statin intolerant, meaning they have side effects that include persistent muscle aches in the arms, shoulders, thighs or hips; weakness; gastrointestinal symptoms; and liver problems.
PSCK9-inhibitors, being developed by rival drug companies Sanofi, Amgen and Regeneron, are antibodies that block the protein PCSK9, thereby helping the body clear LDL cholesterol from the blood. Studies have shown that patients taking the inhibitors have seen their LDL levels drop by as much as half to two-thirds compared to those taking a placebo.
"Here's a way of getting maximal LDL reduction without that problem of statin intolerance," said Dr. Steve Nissen, a Clinic cardiologist who is helping Amgen lead one of their trials to develop evolocumab, their PCSK9-inhibitor.
The FDA is expected to approve the first in this class of drugs next year.
5. Antibody-drug conjugates
Compared to traditional chemotherapy, antibody-drug conjugates (ADCs) are like targeted smart bombs in the fight against cancers.
Antibodies, which by their nature target particular types of tissues, are the perfect vehicle for cancer-fighting drugs to tag along on. By pairing a cancer-fighting drug to an antibody, the drug can do its job in only the place that it's needed, reducing the unnecessary damage to healthy tissues and organs that chemotherapy causes.
Studies have shown that this "bait and switch" strategy works to get the ADC into cancer cells, releases the toxic drug, and kills the cancer cell as it tries to divide.
Some ADCs have already been approved for clinical use-- Kadcyla, a drug for people with HER2-positive metastatic breast cancer, was approved in 2013 -- and more than two dozen are currently in clinical trials for solid tumors and blood cancers.
6. Immune checkpoint inhibitors
Another method of killing cancer cells, by harnessing the body's own immune system, is also showing promise as a treatment for several types of cancer, including melanoma, renal cell carcinoma, and others.
The class of drugs called immune checkpoint inhibitors are administered by IV and block the action of certain proteins in cells that act as inhibitors or "brakes" on the immune system, thereby allowing it to do its job of targeting and killing cancer cells.
Ipilimumab, which was approved in 2011 and was the first immune checkpoint inhibitor available, significantly extended survival for patients with melanoma in clinical trials. Another drug in the class, Keytruda (generic name: pembrolizumab), was approved in September for the treatment of patients with advanced melanoma or melanoma that cannot be surgically treated, and who are no longer responding to other drugs.
Nivolumab, which targets a different checkpoint protein, is currently being tested in clinical trials for the treatment of patients with renal cell carcinoma and other cancers. There are also clinical trials testing the effectiveness of using combinations of checkpoint inhibitors to treat different cancers, particularly in patients who do not respond well to other treatments. In one of these trials -- a small Phase 1 trial testing nivolumab and ipilimumab, more than half of patients had an 80 percent reduction in tumor size.
7. Wireless pacemakers
Pacemakers, which deliver lifesaving jolts of electricity to the heart if the pulse becomes dangerously low or out of rhythm, have traditionally been about the size of a deck of cards, and are implanted in a patient's chest. They are connected to the heart by a long set of wires, which deliver the electrical stimulation.
Those wires can break, or become infected: the risk of infection of the chest incision where the generator is inserted in a traditional surgery is about 2 percent, and dislodgement of the leads happens in about 2 to 3 percent of cases.
The Nanostim pacemaker, developed by Minnesota-based St. Jude Medical, is no bigger than a AAA battery, and can be inserted directly into the heart via a catheter. It's a self-contained unit-- no wires necessary-- so patients avoid a chest incision and the potential dangers they pose. The procedure takes about 15 to 30 minutes to implant.
The device is not yet approved in the U.S., and is currently being tested in the LEADLESS II trial after initial positive results. For now, the wireless pacemaker is only for patients who need "single chamber" pacing -- that is, only one ventricle of the heart needs its rhythm stimulated. These simpler cases make up the minority of pacemaker surgeries today, though doctors expect the technology to expand to more patients.
"From a patient comfort point of view and a safety point of view, it's a nice advance and I think it will help a lot of people," Nissen said.
8. New drugs for IPF (idiopathic pulmonary fibrosis)
Idiopathic pulmonary fibrosis, or IPF, is a progressive lung disease that causes scarring and thickening of tissue, leading to shortness of breath and extreme fatigue. The disease affects about 80,000 mostly middle-aged to older adults in the U.S., and its cause is unknown.
The only treatment for IPF is lung transplant.
Two new drugs -- pirfenidone and nintedanib -- may change that. In a recent Phase 3 trial, pirfenidone reduced disease progression, improved lung function, and reduced the risk of death by 48 percent compared to a placebo. In two Phase 3 studies, nintedanib reduced patients' annual rate of lung function decline by up to 55 percent, compared to a 5 percent decrease for those taking a placebo.
Pirfenidone, being developed by several drug companies, is already available in Europe, Japan and Canada. Nintedanib, which is being developed by Boehringer Ingelheim, has received a breakthrough therapy designation that should help speed its approval as well. The designation is reserved for drugs that treat serious or life-threatening conditions when preliminary evidence indicates the therapy could constitute a major advance compared to what's currently available.
9. Intraoperative radiation therapy for breast cancer
Intraoperative radiation therapy, or IORT, is a procedure in which a patient receives radiation immediately following surgery before leaving the operating room. It's been an option for patients with many types of cancer for years, and surgeons began its limited use in breast cancer patients more than a dozen years ago.
In June, 2010, the journal Lancet published the results of TARGIT-A, an international, randomized clinical trial on the procedure for breast cancer patients, showing that IORT was as effective as standard, whole-breast radiation in preventing a recurrence of breast cancer.
IORT for breast cancer patients usually follows a lumpectomy to remove cancerous tumors. A radiation oncologist inserts a probe that delivers radiation to a targeted area where the tumor was removed, all while the patient is still under anesthesia.
At roughly one-fifth of the cost for standard whole breast radiation, which runs around $10,000, IORT stands to be a boon for the roughly 233,000 people the American Cancer Society estimates will be diagnosed with breast cancer this year.
10. New drug for heart failure (ARNI)
About 5.7 million people in the United States have heart failure, which happens when the heart cannot pump enough blood and oxygen to support other organs. About half of people diagnosed with heart failure die within five years, according to the Centers for Disease Control and Prevention.
The gold standard treatments for heart failure -- ACE inhibitors and beta blockers which expand blood vessels and decrease the heart's workload -- have cut the risk of dying from heart failure in half. But the prognosis is still very poor.
Enter a new drug combination, known as ARNI (for angiotensin-receptor neprilysin inhibitor) which combines an ACE inhibitor with another drug that targets neprilysin, an enzyme involved in many processes in the body.
A clinical trial testing the use of ARNI for heart failure, called Paradigm-HF, was stopped early after it was clear that the drug was overwhelmingly beneficial for patients: It cut the mortality rate by 20 percent, and rate of hospitalizations by 21 percent when compared to standard therapy.
The drug, currently called LCZ696, is being developed by Novartis and is expected to be available next year.
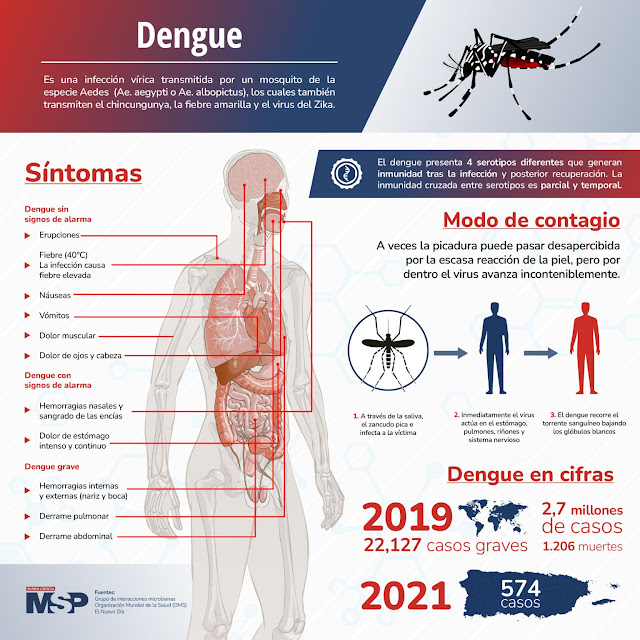
#MSPInfectología | Para la organización Mundial de la Salud (OMS) la incidencia del virus del dengue va aumentando a pasos agigantados a nivel mundial. Según las estimaciones, cada año se reportan alrededor de 390 millones de casos.
La prevención de este virus se puede hacer mediante el manejo adecuado de los desechos sólidos, limpiar y vaciar cada semana los recipientes donde se almacena agua para el uso doméstico y fumigar las zonas estancables y los productos susceptibles de traer el mosquito.
Aprende más sobre este virus con esta infografía o en https://lnkd.in/dSunwEi
LIBROS GRATIS #PDF solo dirígete al enlace de las fotos y allí tienes el enlace en donde puedes descargar de forma legal y sin trucos tu libro. Dr. Ramon Reyes, MD @DrRamonReyesMD https://lnkd.in/eVpFyzZ #DrRamonReyesMD
Blog EMS SOLUTIONS INTERNATIONAL
¿Quién es el Dr. Ramon Reyes, MD?
Blog 8,302,914 visitas por IP https://lnkd.in/daWAMq9
Twitter 422 seguidores
LinkedIn 5451 seguidores
Pinterest 4170 seguidores Mas de 628,000 visitas mensuales
Instagram 3728 seguidores
Facebook 99329 seguidores
Facebook del Blog EMS SSOLUTIONS INTERNATIONAL
62852 seguidores
Grupo de TELEGRAM EMERGENCIAS 4327 miembros
Grupo de TELEGRAM TACMED 613 miembros




No hay comentarios:
Publicar un comentario